Rui Chen
Research Interests:
Silver nanoparticles have been a very attractive material for research because their size is in nanometer range making them a promising candidate for sensor application. Silver nanoparticles absorb visible light and display colors depending on their shape and size. They also react much faster compared to bulk silver. According to the chemical reaction between silver and hydrogen sulfide, we designed an optical sensor based on silver nanoparticles to detect sulfide gas and evaluate its applications in art conservation. Silver nanoparticle film was assembled on glass slide with polyethylenimine binding silver nanoparticles and glass substrate together. The sensor reacts rapidly with sulfide gas and the reaction can be monitored quantitatively by measuring its peak absorption on a photospectrometer. The sensor has been demonstrated to be a sensitive, quantitative, optical sensor for hydrogen sulfide gas.
Silver nanoparticle film substituted for silver foil in Oddy Test
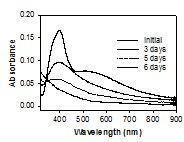
Compared to the conventional Oddy Test, silver nanoparticle films detect sulfide gas emitted in 1 day even the silver foil of the Oddy Test did not show any visible corrosion. Silver nanoparticle films can monitor both the emission of sulfide gas during the test as well as indicate the emission rate of sulfide.

Silver nanoparticle films detect hydrogen sulfide gas at ambient conditions
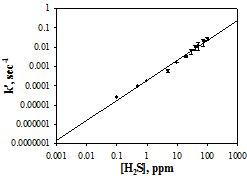
The initial reaction rate of silver nanoparticles with hydrogen sulfide gas (H2S) is demonstrated to be proportional to the gas concentration in part per million by volume. Air, humidity, and volatile compounds such as acetone, ethanol, and ammonia have a negligible effect on the reaction rate, probably including the ambient level of hydrochloric acid. The H2S gas concentration can be determined from either the initial reaction rate or from the H2S dose measured after extended reaction periods. These sensors have been used to study the photodegradation of wool fabrics as well as to examine the condition of daguerreotype photo cases and a silk-lined box.